It seems the assessment of the onset of labour has always been difficult for the observer. Mothers will often report to their clinicians after the event that ‘labour lasted several days’.
We often take this description with a grain of salt and reflect that, although the endpoint is a moment in time, the clinical starting point for labour can be a hard call. Hence a mother’s assessment as the commencement of pain remains a challenge to our clinical judgment and perhaps our compassion too.
Scientific and clinical judgment combine to call a start to labour with a notion of progressive cervical dilatation and the onset of regular, painful uterine contractions; some would add descent of the presenting part. It still leaves us with a diagnosis made over time – a dynamic not a spot diagnosis – at least in many cases. Through history, there have been many innovations and methods to help decide when labour starts, however, the field is still wide open for continued searching for certainty. The pressure for certainty of labour onset comes from a need to measure the effectiveness of various innovations to improve pregnancy outcome and perhaps less from pure scientific inquiry.
Donaldson, working at the City of London Maternity Hospital in 1972, wrote:
‘The lack of progress of measurement in normal labour contrasts with development in other specialties. There is a case for more exact measurement of frequency and strength of uterine contractions and of dilatation of the cervix and, for this purpose, there is a need for new
techniques and approaches, including development of electrical tracings and a plastic cup for obtaining
information from the cervix’.1
This was the time of the accoucheur-mechanic and the march was on to develop such instruments as intrauterine transducers, intrapartum cervical dilators and mechanical, electronic and ultrasound cervimetric devices. The epidural block moved into obstetric parlance and use (obstetric registrars were taught the technique at Crown Street Women’s Hospital, Sydney), thus more readily allowing application of these devices, which were often as ungainly as they were painful. Almost 20 years before, Drew Smythe and Harvey Carey may have managed extraordinarily uncomfortable procedures, but epidurals made intrapartum manipulations more routine in the 1970s. In the US in 1963, researchers wrote of transabdominal insertion of intrauterine transducers2 and by the early 70s Townsend and Francis in the UK had advanced development to a solid-state transducer that was introduced via a plastic, Drew Smythe-like catheter through the cervix after rupturing membranes for induction. An intravenous infusion of oxytocin was commenced and the rate of infusion directed by the intrauterine pressure and contraction frequency as recorded by the transducer. Thus there developed a potential and actual closed system, with the unfortunate midwife as an onlooker and machine operator in the quest for uterine efficiency and reduction of perinatal morbidity and mortality. This was the way in 1975, in many UK hospitals, when primipara were induced on the dot of 38 weeks if they had shown any BP readings over 120/80. 24-hour induced labour abounded but the caesarean section rate was kept to seven per cent by ‘efficient’ acceleration of the oxytocin infusion – safety being guarded by the addition of a scalp electrode – now John Cleese and Monty Python were truly ascendant.
As recently as January 2010, an article from Amsterdam in the New England Journal of Medicine suggests that expectations for the superiority of internal over external tocometry may be overstated when it concludes that:
‘Internal tocodynamometry during induced or augmented labour, as compared with external monitoring, did not significantly reduce the rate of operative deliveries or of adverse neonatal outcomes.’3
Yet still, there was an instrument-in-waiting. In 1976, Richardson et al wrote of the development of an instrument for monitoring dilatation of the cervix during labour. It literally had bulldog clips, being described as ‘non-traumatic, accurate and easy to apply’. With this instrument, it was found that ‘the cervix opens in an oscillatory manner dictated by the strength and period of uterine contractions’.4
Similar instruments formed the basis of the early ultrasound methods also; they relied on:
‘…two piezoelectric crystals attached on the uterine cervix. A small spring-loaded clip allowed each crystal to be
fixed on the rim of the cervical os. Clinical accuracy was +/- 0.6 cm. When the ultrasound recording of cervical dilatation is compared to the intrauterine pressure curve, it is characterised by a baseline and wave-shape curve of dilatation (DWP).’ 5
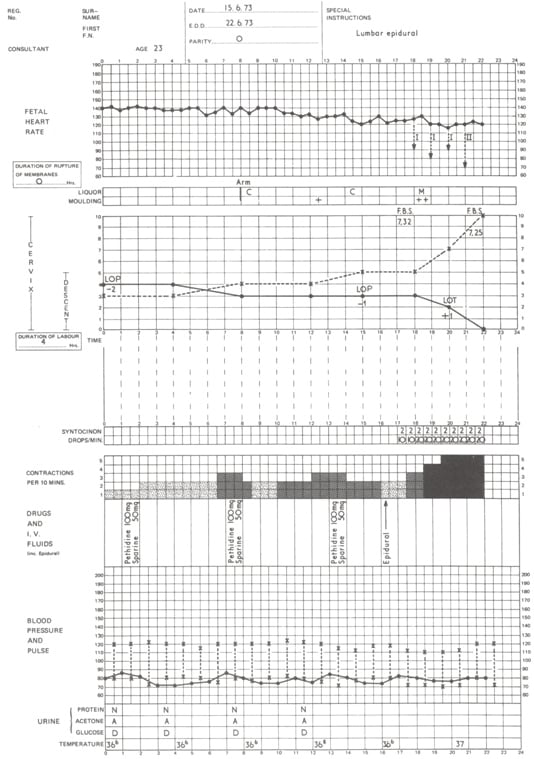
Figure 1. Partogram showing annotation of events in patient with prolonged labour. (From Studd, op. cit.)
By 1995, transperineal6 ultrasound was said to be an appropriate tool for demonstrating progressive effacement and dilatation at the beginning of labour in term pregnancies. Sonography of the cervix in early labour, before during and after a uterine contraction was described7, by Saito et al at the University of Tokyo, as an effective tool for assessment in early labour. They concluded that they could differentiate between inefficient and normal uterine contractions and thus predict the subsequent course of labour. Using transvaginal sonography, Swedish researchers Strobel et al showed that, in prolonged pregnancy, the Bishop score and sonographic assessment of cervical length had similar ability in predicting time to the onset of labour and delivery. Use of both together is likely to have a synergistic effect.8
In 1954, Friedman developed a graphical representation of cervical dilatation and fetal station against hours from the onset of labour. A typical sigmoid-shaped curve defined the limits of what was considered normal time-related cervical dilatation and an hyperbolic curve described fetal station over time. Eighteen years later, Philpott, in then Rhodesia, brought the alert and action lines – the latter four hours to the right of the former and both sloped at 1 cm of dilatation per hour. Studd added stencils and commenced further clinical research into the partogram in the mothers of Birmingham, so that in 1975 he wrote:
‘The partogram is a major advance in modern obstetrics, being appropriate for all labours. In our prospective study of spontaneous labour, the recognition of high-risk labour was aided by the routine use of the labour stencil for both primigravid and multigravid labours. Stimulation of labour solves far more problems than it creates, but it is essential that careful selection of patients should take place to
prevent this major obstetric advance being abused and discredited.’ 9
The Proceedings of the Royal Society of Medicine in 1972 were full of articles by such contemporary luminaries as Kieran O’Driscoll of the Rotunda, R H Philpott of the University of Rhodesia, J W W Studd of Birmingham Maternity Hospital, J M Beazley of Queen Charlotte’s Maternity Hospital, R W Beard of King’s College Hospital and C P Douglas of the Royal Free Hospital. These articles dealt with the use of the partogram in labour and the place of augmentation with oxytocin. Beazley argued that:
‘Partograms of the Queen Charlotte’s type facilitate:
- Adequate anticipation of prolonged labour;
- Standardisation of abnormalities in a reproducible manner; and
- Distinctions between minimal risk and hazardous labours.
They cost very little and improve obstetric management without disturbing normal labour ward routines.’
Beard, accepting O’Driscoll’s finding that oxytocin rendered even regular strong contractions more efficient, cautioned that it had been: ‘…clearly demonstrated that the mean pH of fetuses delivered after oxytocin-stimulated labour was significantly lower than that of fetuses delivered after spontaneous labour’.10 He advised increased surveillance of these augmented labours.
Current research on instrumentation includes evaluation of cervical compliance. In 1989, Brundin and colleagues from the Karolinska Institute, Sweden, measured cervical tissue resistance with a strain guage and cervical dilators, recording resistance on a polygraph. They suggested peak resistance provided the most accurate measurement of human cervical resistance.11 Alongside this physical appraisal, further research highlighted the biochemical changes in cervical tissue in pregnancy and towards term. Aubard, of the Service de Gynécologie-Obstétrique, CHU Limoges, wrote that the gravid cervix is strikingly less muscular than the rest of the uterus and that cervical connective tissue is largely collagen fibres and a matrix ‘rich in proteoglycans’. He goes on to observe how the collagen fibres ‘dissolve’ shortly before labour and this ‘maturation’ is perhaps mediated through steroid hormones, prostaglandins and collagenases.12
In 2003, Frey wrote of ‘real-time elastography’13 and the sonographic techniques that might be enjoined to assess the alteration in ‘tissue hardness’ of the cervix. Initially seen as a method of cancer investigation in more superficial tissues, this modality is now being investigated for its potential to portray the changes in tissue resistance in the cervix towards term and in early labour. The basic physics of elastography is that tissue compression produces strain (displacement) in the tissue under evaluation and that the firmer the tissue, the less the strain. It is anticipated this may give early warning of the chance of less-than-adequate progress in labour and thus earlier intervention. Thomas14 from the University of Berlin, has taken this idea forward, particularly in the search to define the at-risk group for premature delivery and in so doing, may well be outlining a simple ultrasound method (based on colours too!) to assess a starting point for labour and an early labour analysis of chances of vaginal delivery.
As well as its potential for assistance for early labour cervical assessment, ultrasound may have a place in assessing the progress of the descent of the fetus and in confirmation of the position of the head at end of first stage and during delivery – we have all delivered mothers of a baby in an unexpected occiput posterior position. Molina and Nicolaides write that digital examination of fetal head position prior to instrumental delivery has a high proportion of incorrect calls on the position.15 Certainly, too, if you are still performing twin deliveries vaginally, ultrasound helps with position assessment of the second twin. (Halloway, personal communication!)
So shall we look to our patients’ welfare and fulfillment for the final decision on the use of tools and instruments to help with labour assessment? Midwives are sometimes the recipients of our dissatisfaction when second stage ends up going for three or more hours, but what dysfunction might we be causing to their ability to care for their patients if we cause them to be the mechanics at delivery by introduction of unproven ‘instruments of labour’. We should be ever alert to the evidence base – at least in this area!
References
- I A Donaldson. Proc R Soc Med. 1972 October; 65(10): 891.
- Med Electron Biol Engng. 1964; Vol 2, p173-178, Pergamon Press. Printed in Great Britian.
- Bakker, et al. N Engl J Med. 2010; 362:306-13.
- Richardson, et al. Biomed Eng. 1976 Sep;11(9):311-3.
- Moss P L. Am J Obstet Gynecol. 1978 Sep 1;132(1):16-9.
- Zilanti M, et al. J Ultrasound Med. 1995 Oct; 14 (10):7 19-24.
- Saito M, et al. Ultrasound Obstet Gynecol. 2003 Dec; 22(6):604-8.
- Strobel E, et al. Ultrasound Obstet Gynecol. 2006 Sep; 28(3):298-305.
- Studd, et al. BMJ 1975; 2:545-547.
- op cit, Proc R Soc Med.
- Brundin, et al. Journal of Biomedical Engineering 1989 Mar; 11(2):148-150.
- Aubard. J Gynecol Obstet Biol Reprod (Paris).1998 Dec; 27(8):755-64.
- Frey, et al. Radiologe. 2003 Oct; 43(10):850-5.
- Thomas A. Ultrasound Obstet Gynecol. 2006; 28:356-357.
- Molina and Nicolaides. Fetal Diagn Ther. 2010; 27(2):61-7. Epub 2010 Feb 18.






Leave a Reply