While textbooks contain little information on pelvic floor trauma in childbirth, it is clear that many women giving birth to their first child vaginally suffer a degree of permanent damage to their pelvic floor structures. For example, anal sphincter trauma is much more common than generally assumed1,2, frequently not recognised in the delivery suite1,2 and often is suboptimally repaired, even in tertiary units.3 The resulting defects of internal and external anal sphincter muscle are the main aetiological factor for faecal incontinence in later life.4
Similarly, it is now clear that pelvic floor trauma encompasses more than what we were taught to identify in the delivery suite. In about half of all women after vaginal delivery there is substantial alteration of functional anatomy affecting the puborectalis component of the levator ani muscle.5 The integrity of this structure is currently the best-defined aetiological factor in the pathogenesis of prolapse, a condition for which 10–20 per cent of all women will eventually undergo surgery at least once in their lifetime.6,7
The levator ani muscle is part of the abdominal envelope, a muscular plate surrounding a central v-shaped ‘levator hiatus’, which encloses the urethra, vagina and anorectum. It necessarily is a compromise between conflicting priorities: the abdominal organs have to be secured against the pressure differential between inside and outside and against gravity, solid and liquid wastes have to be evacuated in a controlled, socially acceptable fashion and babies are supposed to pass through the levator hiatus, which is a particular challenge in view of the size of the baby’s head.
Figure 1 shows the levator ani as seen from caudally, in a fresh cadaver on the left, and in an asymptomatic volunteer on pelvic floor 3D ultrasound on the right. The puborectalis muscle is evident as a v-shaped structure between 5mm and 10mm in thickness, anchored to the inferior pubic rami and the body of the os pubis on both sides.
Both the external and internal anal sphincter are commonly impacted by childbirth owing to their location within the levator hiatus, in immediate proximity to the fetal head on crowning. The anal canal is a tubular structure of 3–5cm in length, with a smooth muscle tube (the internal anal sphincter [IAS]) surrounded in its distal half by a donut-shaped structure of striated muscle (the external anal sphincter [EAS]), see Figure 2 for a tomographic representation in a nulliparous patient. The anal canal undergoes marked displacement and distortion during childbirth. It is protected to some degree by the pyramidal fibromuscular structure of the perineal body, but as this is commonly disrupted during a first vaginal delivery, this protection is rather incomplete.
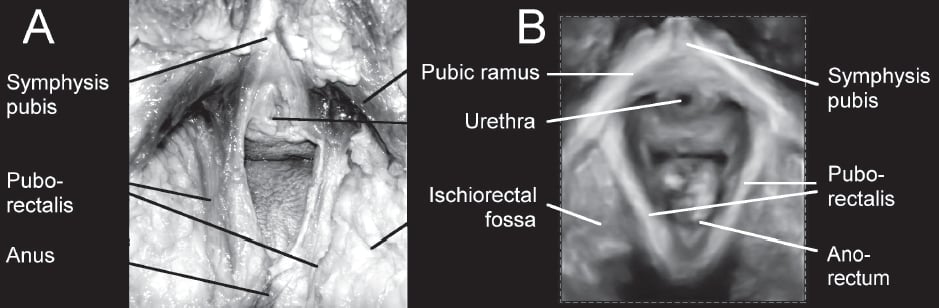
Figure 1. A. Intact puborectalis muscle in a fresh cadaver, dissected from caudally. The vulva, mons pubis, clitoris, perineal muscles and perineum to the anus, as well as peri- and postanal skin and the fibrofatty tissue of the ischiorectal fossa have been removed to allow access to the puborectalis muscle. B. The appearance of the puborectalis muscle in a rendered volume in the axial plane, using translabial 3D ultrasound.
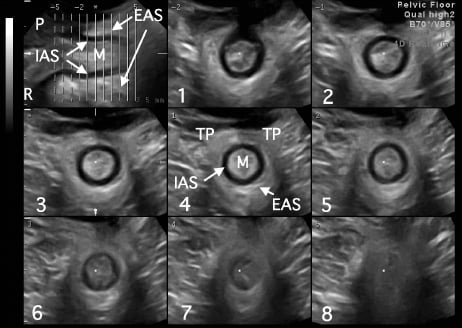
Figure 2. Normal appearances of the anal canal in a nulliparous patient as seen on translabial tomographic ultrasound. The top left image shows the midsagittal plane, the remaining eight slices are placed from just above the EAS (slice 1) to just below the IAS (slice 8), encompassing the entire EAS. Reproduced with permission.2
In the past, episiotomy was employed to prevent extension of a perineal midline tear into the anal sphincter. It is commonly assumed that episiotomy does not protect the anal sphincter8, but all studies in this field to date have used clinical diagnosis as an outcome measure. Since we now know that the clinical diagnosis of sphincter tears is often inadequate1, 2, this issue will have to be revisited. It is conceivable that the association between episiotomy and sphincter tears seen in past trials is a detection artefact, owing to a higher likelihood of correct diagnosis in women in whom tissues were exposed by an episiotomy.
Trauma to the levator ani
The levator ani muscle plays a major role in childbirth as it is the major soft tissue structure defining the dimensions and biomechanical properties of the vagina.9 During a vaginal delivery it has to undergo substantial distension.10,11 Data from muscle physiology research suggests that skeletal muscle will not stretch to more than twice its length without suffering ultrastructural or macroscopic trauma.12 It is therefore remarkable that in about half of all women there is no appreciable alteration in distensibility or morphological appearance after vaginal childbirth, and we assume that this is somehow owing to the protective hormonal effects of pregnancy.
The commonest form of macroscopic levator trauma is an avulsion: a traumatic dislodgment of the puborectalis muscle from its bony insertion (see Figures 3 and 4). This has a substantial impact on hiatal dimensions.13 In addition, there is irreversible overdistension of the levator hiatus in more than a quarter of all women after vaginal childbirth.5 Excessive distensibility of the hiatus (‘ballooning’), whether as a result of childbirth or congenital, is associated with prolapse14 and prolapse recurrence.15 Ballooning and avulsion, while often associated, seem to be independent predictors of prolapse and prolapse recurrence.15, 16 Appearances are rarely consistent with pudendal neuropathy, which in the past was considered the main aetiological factor in pelvic floor dysfunction17, 18, even in women after severely obstructed labour.19
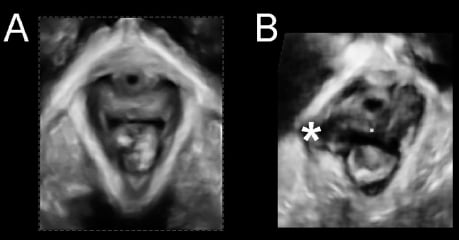
Figure 3. A comparison of ultrasound findings in a normal patient (A) and in a patient with a typical right-sided avulsion (B); rendered volume, axial plane, indicated by a ‘*’. It is evident that the morphological abnormality documented here is an ‘avulsion’ of the puborectalis muscle insertion, ie., the muscle has separated from its bony insertion.
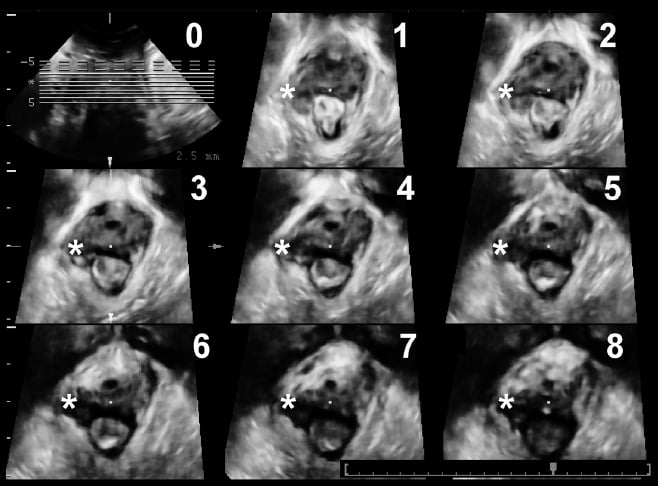
Figure 4. Assessment of the puborectalis muscle by tomographic or multislice ultrasound. Image 0 represents a reference image in the coronal plane. Images 1–8 show slices parallel to the plane of minimal hiatal dimensions. Slices 1 and 2 are 5 and 2.5mm below this plane, slice 3 represents the plane of minimal dimensions and slices 4–8 are 2.5–12.5mm above this plane, likely encompassing the entire insertion of the puborectalis. Reproduced with permission.20
Avulsion is most commonly diagnosed by tomographic ultrasound20 (see Figure 4) or magnetic resonance imaging (MRI)21, but it is palpable vaginally.22-25 It requires some instruction, but diagnosis by palpation can very likely be as valid as diagnosis by imaging.26 The index finger is placed parallel to the urethra, with the tip of the finger at the bladder neck and its palmar surface adjacent to the posterior/dorsal surface of the os pubis. An intact muscle leaves just enough room to fit the palpating finger between urethra medially and insertion of the puborectalis muscle laterally. If there is no muscle palpable on the os pubis and its inferior ramus immediately lateral to a finger placed parallel to the urethra and if this finger can be moved over the inferior pubic ramus without encountering muscle for 2–3cm, a diagnosis of levator avulsion is made. Poor contraction strength can help alert the examiner to an increased likelihood of avulsion25, and there are a number of other predictors.27
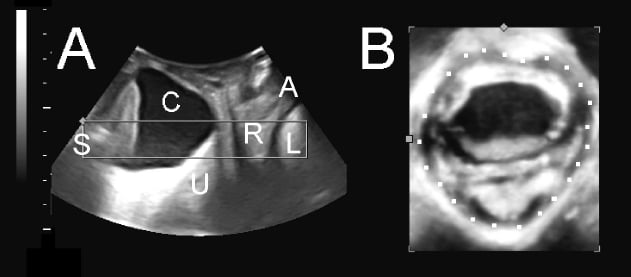
Figure 5. Typical findings in a patient with a third-degree cystocele (indicated with a ‘C’ in image A), bilateral avulsion and marked hiatal ballooning, the latter visible in the axial plane in B, with a dotted line illustrating the hiatus. S = symphysis pubis, C = cystocele, U = uterus, R = rectal ampulla, A = anal canal.
Avulsion often causes marked asymmetry of the hiatus28 and sometimes this asymmetry is evident even on clinical inspection. On Valsalva, the anus and perineum are displaced towards the healthy side, pushed by a prolapse descending preferentially on the avulsed side. On pelvic floor muscle contraction, the perineum and anus are pulled by the contralateral intact muscle, again resulting in displacement towards the healthy, undamaged side.
While about 10–30 per cent of women will suffer macroscopic levator trauma5,29-35, there is an even greater number who sustain ‘microtrauma’, in other words irreversible overdistension of the levator hiatus.5 The predictors of microtrauma may vary from those that predict levator avulsion.5 It is not yet clear what the long-term impact of such morphological and functional changes is, but neither ongoing deterioration nor ‘healing’ is likely to be common.36
It is likely that factors such as birthweight, length of the second stage, size of the fetal head and forceps delivery increase the probability of avulsion injury.5, 29-35,37 However, such ‘predictors’ are of very limited use since they are not available before the onset of labour. In order to prevent levator avulsion, we would need predictors that can be determined during pregnancy. In some studies, avulsion seems associated with maternal age at first delivery.29, 38, 39 Finally, there is ample evidence suggesting it is usually the first vaginal delivery that causes by far the most morphological and functional alteration, both in terms of actual tears as well as in terms of levator distensibility or pelvic organ support.40-43
Traumatic disconnection of the levator ani from its insertion has substantial consequences for function. Contraction strength as estimated by Oxford grading25 and instrumented speculum44 is reduced by about a third, an observation that may help diagnose levator trauma. Avulsion results in a hiatus that is larger (by 20–30 per cent), especially in the coronal plane45, more distensible and less contractile.44,13 Most importantly, avulsion is associated with symptoms and signs of prolapse.23, 25 Women seem to notice the effect of avulsion as a reduction in contraction strength on pelvic floor muscle contraction46,47 and as increased vaginal laxity and reduced tone on intercourse.48
In the medium to long term, levator avulsion is associated with anterior and central compartment prolapse and likely represents at least part of the missing link between childbirth and prolapse.49 The larger a defect is, both in width and depth, the more likely are symptoms and/or signs of prolapse.50 Levator avulsion seems to markedly increase the risk of significant anterior and central compartment prolapse.51 Figure 5 shows typical findings in a patient with bilateral avulsion, marked hiatal ballooning and a third-degree cystocele.
The most important issue for clinical practice is that both avulsion and ballooning seem to be risk factors for prolapse recurrence both on ultrasound51-54 and on MRI.55 This implies that such findings should be obtained preoperatively and are likely to be useful for surgical planning. Use of anchored anterior compartment mesh may partly compensate for the increased recurrence risk conveyed by avulsion.56 Diagnosing levator avulsion and/or hiatal ballooning may not require imaging. Both avulsion20 and ballooning (via ‘genital hiatus’ [gh] and ‘perineal body’ [pb] measurements included in the International Continence Society Pelvic Organ Prolapse Quantification System [POP-Q] system) can be diagnosed clinically, and we have determined a cut-off of 7cm for the sum of gh and pb to define ballooning clinically.57, 58
Avulsion does not seem to be associated with stress urinary incontinence (SI) and urodynamic stress incontinence (USI)59,60, and there is conflicting evidence as regards fecal incontinence.61,62,63 Sexual function may also be impacted, especially owing to reduced vaginal tone or ‘vaginal laxity’, but there is little information on this issue so far.
Trauma to EAS and IAS
EAS and IAS trauma is much more common than generally assumed, and commonly missed in the delivery suite. Obstetric anal sphincter injuries (OASIS) are considered an important risk factor for faecal incontinence and are commonly identified among women with anal incontinence in later life.4 The reported incidence of OASIS varies between 0.5 per cent and five per cent64, 65, which is clearly an underestimate.66 Up to 35 per cent of primiparous women have been found to show evidence of OASIS on transanal ultrasound at six weeks postpartum.67 A meta-analysis of 717 vaginal deliveries showed an incidence of anal sphincter defects in 26.9 per cent of primiparous women68 on endo-anal ultrasound, and own work using transperineal exo-anal 3D/4D ultrasound demonstrated significant EAS defects in 28 per cent of women after a first vaginal delivery. Of such defects, 87 per cent were undiagnosed clinically.2 This may be either owing to missed diagnosis or because of clinically occult trauma.1, 69-71 Either way, it is evident that the assessment of anal sphincters requires imaging, since clinical data are of very limited utility.
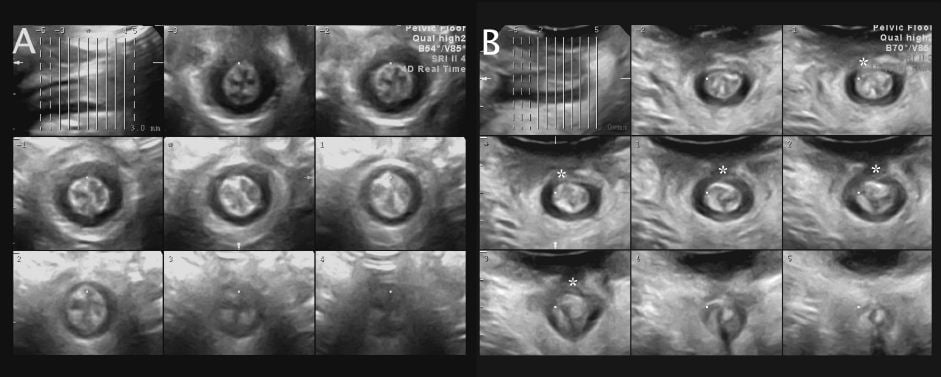
Figure 6. Anal sphincter at 36 weeks gestation (a) and after normal vaginal delivery at term, with a second-degree perineal tear documented in the delivery suite. There is a complete defect of the external anal sphincter (*) (b). The top left image in both parts shows the anal canal in the midsagittal plane. The rest are coronal slices through the anal canal at 3mm interslice intervals. Reproduced with permission.57
The anal sphincters are commonly evaluated with endoanal ultrasound, which is regarded as the gold standard. However, this technique is not universally available. In addition, endoanal imaging is invasive, involving the insertion of an ultrasound probe into the anal canal. This distorts anatomy and precludes dynamic evaluation of the anal sphincter and mucosa on sphincter contraction which seems to enhance the definition of muscular defects.72 The method is probably not suitable for routine imaging after childbirth, and this has hampered translation into clinical practice. Exo-anal or transperineal ultrasound73,74, on the other hand, does not have these disadvantages and is increasingly used to evaluate the anal sphincters. A recent study comparing 3D transperineal ultrasound and 2D endo-anal ultrasound in the detection of anal sphincter defects has shown good agreement between the two techniques.75 One major advantage of the exo-anal method is that it is easily combined with imaging of other pelvic floor structures such as the levator ani muscle. Figure 6 shows a tomographic representation of normal EAS and IAS at 36 weeks and an obvious EAS defect three months after a normal vaginal delivery at term, with only a second degree tear documented at birth.
Clinical consequences
Informed consent
First and foremost, we have a legal obligation that arises from the patient’s right to know of material risks, a right which in turn arises from the patient’s right to decide whether or not to submit to the medical treatment proposed.76 There can be no doubt that maternal birth trauma is a common enough risk (affecting 30–50 per cent of all primiparae after a vaginal delivery) to require disclosure.
Maternity services performance
The fiduciary duty we have regarding patient care also implies that we need to provide information to policymakers and health services. We need to promote the use of maternal pelvic floor trauma as a performance indicator of maternity services, since it has potential consequences for future life, even if it may take decades for clinically overt conditions to develop.77 As clinical diagnosis is insufficient, assessment of maternal birth trauma may require imaging two or more months after childbirth. Such a postnatal service would not just allow the noninvasive diagnosis of levator trauma and OASIS, but would also provide an opportunity to discuss issues not commonly given a high priority in the immediate postpartum period, such as contraception and sexual health.78 It would also allow for a debriefing after traumatic childbirth and enable earlier recognition of postnatal depression, a condition that is one of the top causes of maternal childbirth-related mortality.79
Prevention
An immediate clinical consequence should be to discourage forceps delivery. Forceps (as opposed to vacuum) is a modifiable risk factor for both levator avulsion and OASIS and should be avoided if possible. While perineal protection may have some value for the prevention of anal sphincter trauma, all data currently available use clinical diagnosis as main outcome measure and are therefore of very limited utility.
As levator avulsion and obstetric anal sphincter trauma are exclusively caused by vaginal childbirth, elective caesarean delivery would be expected to completely prevent such trauma. On the current state of the literature, it would be difficult to defend a policy that denies women the right to elective caesarean delivery on maternal demand. However, caesarean section has substantial disadvantages, both for mother and infant, which are beyond the scope of this paper. Hence, other forms of prevention may be more practicable, such as attempts to change the biomechanical properties of the birth canal.
There is a commercially available device, the Epi-No™, that is used to dilate the perineum and vagina in the last few weeks of pregnancy, and this device has been shown to reduce perineal trauma.80 The Epi-No is currently under investigation regarding a potential role in pelvic floor protection in the unit of the author, and results of a pilot study are promising.81 There are several other interventions that could be tested for a potential protective role. Since the degree of trauma sustained by skeletal muscle on stretching is directly proportional to the amount of work needed to elongate the muscle,12 it seems reasonable to expect that muscle paralysis (via a pudendal nerve block or a dense epidural) might effect a reduction of trauma. We have commenced a pilot randomised controlled trial to test such a hypothesis.
Conclusions
Urogynaecology is undergoing a period of rapid change and development owing to the recent introduction of modern functional imaging. The most marked impact of this change in our diagnostic capabilities has been on the issue of childbirth-related pelvic floor trauma. It is likely that women will increasingly demand action on this count as they become aware of the risks that vaginal childbirth poses to their future health.
Editorial comment
There is no doubt that perineal trauma during vaginal delivery is a hot topic. The development of functional imaging techniques has revolutionised our understanding of the pelvic floor during and after birth. We are fortunate to have one of the foremost international experts in this field as a Fellow of our College.
Those who have been to a College scientific meeting in the last few years will likely have heard Prof Dietz speak on this topic. In many ways, the message he has for us can be considered confronting. With this in mind, we have invited Peter to encapsulate his message and conclusions for the readers of O&G Magazine. As always, we hope this article will stimulate discussion and debate and we look forward to hearing from our readership.
A/Prof Steve Robson, O&G Magazine Advisory Committee
References
- Andrews A, Sultan A, Thakar R, Jones P. Occult anal sphincter injuries- myth or reality? BJOG. 2006;113:195-200.
- Guzman Rojas R, Shek K, Langer S, Dietz H. Prevalence of anal sphincter injury in primiparous women. Ultrasound Obstet Gynecol. 2013;42:461-466.
- Shek K, Guzman Rojas R, Dietz HP. Residual defects of the external anal sphincter are common after OASIS repair. Neurourol Urodyn. 2013;31:913-14.
- Thakar R, Sultan A. Anal endosonography and its role in assessing the incontinent patient. Best Practice & Research Clinical Obstetrics & Gynaecology. 2004;18:157-173.
- Shek K, Dietz H. Intrapartum risk factors of levator trauma. BJOG. 2010;117:1485–1492.
- Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL. Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol. 1997;89:501-506.
- Smith F, Holman D, Moorin R, Tsokos N. Lifetime Risk of Undergoing Surgery for Pelvic Organ Prolapse. Obstet Gynecol. 2010;116:1096-1100.
- Carroli G, Mignini L. Episiotomy for vaginal birth. Cochrane Database of Systematic Reviews 2009.
- Jung S, Pretorius D, Padda B, et al. Vaginal high-pressure zone assessed by dynamic 3-dimensional ultrasound images of the pelvic floor. Am J Obstet Gynecol. 2007;197:52.e1-7.
- Lien KC, Mooney B, Delancey JO, Ashton-Miller JA. Levator ani muscle stretch induced by simulated vaginal birth. Obstet Gynecol. 2004;103:31-40.
- Svabik K, Shek K, Dietz H. How much does the levator hiatus have to stretch during childbirth? BJOG. 2009;116:1657-1662.
- Brooks S, Zerba E, Faulkner J. Injury to muscle fibres after single stretches of passive and maximally stimulated muscle in mice. Journal of Physiology. 1995;488:459-469.
- Abdool Z, Shek K, Dietz H. The effect of levator avulsion on hiatal dimensions and function. Am J Obstet Gynecol. 2009;201:89.e1-89. e5.
- Dietz H, De Leon J, Shek K. Ballooning of the levator hiatus. Ultrasound Obstet Gynecol. 2008;31:676-680.
- Rodrigo N, Shek K, Wong V, Martin A, Dietz H. Hiatal ballooning is an independent risk factor of prolapse recurrence. Int Urogynecol J. 2012;23:S 129-130.
- Dietz H, Franco A, Shek K, Kirby A. Avulsion injury and levator hiatal ballooning: two independent risk factors for prolapse? An observational study. Acta Obstet Gynecol Scand. 2012;91:211-214.
- Swash M, Snooks SJ, Henry MM. Unifying concept of pelvic floor disorders and incontinence. Journal of the Royal Society of Medicine. 1985;78:906-911.
- Allen RE, Hosker GL, Smith AR, Warrell DW. Pelvic floor damage and childbirth: a neurophysiological study. BJOG. 1990;97:770-779.
- Dietz H, Habtemariam T, Williams G. Does obstructed labour in women with urogenital fistula lead to atrophy of the levator ani muscle? J Urol. 12;188:1772-7.
- Dietz H, Bernardo M, Kirby A, Shek K. Minimal criteria for the diagnosis of avulsion of the puborectalis muscle by tomographic ultrasound. Int Urogynecol J. 2011;22:699-704.
- Zhuang R, Song Y, Chen Q, et al. Levator avulsion using a tomographic ultrasound and magnetic resonance–based model. Am J Obstet Gynecol. 2011;205:232.e1-8.
- Dietz HP, Hyland G, Hay-Smith J. The assessment of levator trauma: A comparison between palpation and 4D pelvic floor ultrasound. Neurourol Urodyn. 2006;25:424-427.
- Kearney R, Miller JM, Delancey JO. Interrater reliability and physical examination of the pubovisceral portion of the levator ani muscle, validity comparisons using MR imaging. Neurourology & Urodynamics. 2006;25:50- 54.
- Dietz HP, Shek KL. Validity and reproducibility of the digital detection of levator trauma. Int Urogynecol J. 2008;19:1097-1101.
- Dietz HP, Shek C. Levator Avulsion and Grading of Pelvic Floor Muscle Strength. Int Urogynecol J. 2008;19:633-636.
- Dietz H, Moegni F, Shek K. Diagnosis of Levator avulsion injury: a comparison of three methods. Ultrasound Obstet Gynecol. 2012;40:693-698.
- Dietz H, Kirby A. Modelling the likelihood of levator avulsion in aurogynaecological population. ANZJOG. 2010;50:268-272.
- Dietz H, Bhalla R, Chantarasorn V, Shek K. Avulsion of the puborectalis muscle causes asymmetry of the levator hiatus. Ultrasound Obstet Gynecol. 2011;37:723-726.
- Dietz H, Lanzarone V. Levator trauma after vaginal delivery. Obstet Gynecol. 2005;106:707-712.
- Krofta L, Otcenasek M, Kasikova E, Feyereisl J. Pubococcygeus-puborectalis trauma after forceps delivery: evaluation of the levator ani muscle with 3D/4D ultrasound. Int Urogynecol J. 2009;20:1175-1181.
- Albrich S, Laterza R, Skala C, Salvatore S, Koelbl H, Naumann G. Impact of mode of delivery on levator morphology: a prospective observational study with 3D ultrasound early in the postpartum period. BJOG. 2012;119:51-61.
- Valsky Dv, Lipschuetz M, Bord A, et al. Fetal head circumference and length of second stage of labor are risk factors for levator ani muscle injury, diagnosed by 3-dimensional transperineal ultrasound in primiparous women. Am J Obstet Gynecol. 2009;201:91.e1-91.e7.
- Blasi I, Fuchs I, D’Amico R, et al. Intrapartum translabial threedimensional ultrasound visualization of levator trauma. Ultrasound Obstet Gynecol. 2011;37:88-92.
- Cassado Garriga J, Pessarodona Isern A, Espuna Pons M, Duran Retamal M, Felgueroso Fabregas A, Rodriguez- Carballeira M. Tridimensional sonographic anatomical changes on pelvic floor muscle according to the type of delivery. Int Urogynecol J. 2011;22:1011-1018.
- 35 Chan S, Cheung R, Yiu A, et al. Prevalence of levator ani muscle injury in Chinese primiparous women after first delivery. Ultrasound Obstet Gynecol. 2012;39:704-709.
- Shek K, Dietz H. Does Levator Trauma heal? Int Urogynecol J 2011;22:S12-13.
- Kearney R, Fitzpatrick M, Brennan S, et al. Levator ani injury in primiparous women with forceps delivery for fetal distress, forceps for second stage arrest, and spontaneous delivery. Int J Gynaecol Obstet. 2010;111:19-22.
- Kearney R, Miller J, Ashton-Miller J, Delancey J. Obstetric factors associated with levator ani muscle injury after vaginal birth. Obstet Gynecol. 2006;107:144-9.
- Dietz H, Simpson J. Does delayed childbearing increase the risk of levator injury in labour? ANZJOG. 2007;47:491-495.
- Dietz HP, Clarke B, Vancaillie TG. Vaginal childbirth and bladder neck mobility. ANZJOG. 2002;42:522-525.
- Horak A, Guzman Rojas R, Shek K, Dietz H. Pelvic Floor Trauma: does the second baby matter? Int Urogynecol J. 2012;23:S175-176.
- Dickie K, Shek K, Dietz H. The relationship between urethral mobility and parity. BJOG. 2010;117:1220-1224.
- Kamisan Atan I, Shek K, Gerges B, Dietz H. The association between vaginal childbirth and hiatal dimensions. IUGA 2012. Brisbane, 2012.
- Delancey J, Morgan D, Fenner D, et al. Comparison of levator ani muscle defects and function in women with and without pelvic organ prolapse. Obstetrics & Gynecology. 2007;109:295-302.
- Otcenasek M, Krofta L, Baca V, et al. Bilateral avulsion of the puborectal muscle: magnetic resonance imaging-based three-dimensional reconstruction and comparison with a model of a healthy nulliparous woman. Ultrasound in Obstetrics & Gynecology. 2007;29:692-696.
- Dietz H, Shek K, Chantarasorn V, Langer S. Do women notice the effect of childbirth-related pelvic floor trauma? ANZJOG. 2012;DOI 10.1111/j.1479-828X.2012.01432.x.
- Thibault-Gagnon S, Yusuf S, Langer S, Wong V, Shek K, Dietz H. Do women notice the impact of childbirth-related tevator trauma on pelvic floor and sexual function? Int Urogynecol J. 2014; in print.
- Thibault-Gagnon S, Yusuf S, Langer S, Wong V, Shek K, Dietz H. Do women notice the impact of childbirth-related tevator trauma on pelvic floor and sexual function? Int Urogynecol J. 2012;23:S183-185.
- Dietz H, Simpson J. Levator trauma is associated with pelvic organ prolapse. BJOG. 2008;115:979-984.
- Dietz H. Quantification of major morphological abnormalities of the levator ani. Ultrasound Obstet Gynecol. 2007;29:329-334.
- Dietz HP, Chantarasorn V, Shek KL. Levator avulsion is a risk factor for cystocele recurrence. Ultrasound Obstet Gynecol. 2010;36:76-80.
- Model A, Shek Kl, Dietz HP. Levator defects are associated with prolapse after pelvic floor surgery. Eur J Obstet Gynecol Reprod Biol. 2010;153:220-223.
- Weemhoff M, Vergeldt T, Notten K, Serroyen J, Kampschoer P, Roumen F. Avulsion of puborectalis muscle and other risk factors for cystocele recurrence: a 2-year follow-up study. Int Urogynecol J. 2012;23:65-71.
- Wong V, Shek K, Goh J, Rane A, Dietz HP. Is levator avulsion a predictor for cystocele recurrence following anterior compartment mesh? Neurourol Urodyn. 2011;30:879-880.
- Morgan D, Larson K, Lewicky-Gaupp C, Fenner D, Delancey J. Vaginal support as determined by levator ani defect status 6 weeks after primary surgery for pelvic organ prolapse. Int J Gynaecol Obstet. 2011;114:141-144.
- Rodrigo N, Wong V, Shek K, Martin A, Dietz H. The use of 3-dimensional ultrasound of the pelvic floor to predict recurrence risk after pelvic reconstructive surgery. ANZJOG. 2014; in print.
- Khunda A, Shek K, Dietz H. Can ballooning of the levator hiatus be determined clinically? Am J Obstet Gynecol. 2012;206:246.e1-4.
- Gerges B, Kamisan Atan I, Shek K, Dietz H. How to determine ‘ballooning’ of the levator hiatus on clinical examination. Int Urogynecol J. 2012;23:S52- 53.
- Dietz H, Kirby A, Shek K, Bedwell P. Does avulsion of the puborectalis muscle affect bladder function? Int Urogynecol J. 2009;20:967-972
- Morgan D, Cardoza P, Guire K, Fenner D, Delancey J. Levator ani defect status and lower urinary tract symptoms in women with pelvic organ prolapse. Int Urogynecol J. 2010;21:47-52.
- Chantarasorn V, Shek K, Dietz H. Sonographic detection of puborectalis muscle avulsion is not associated with anal incontinence. ANZJOG. 2011; 51:130-135.
- Van De Geest L, Steensma AB. Three-dimensional transperineal ultrasound imaging of anal sphincter injuries after surgical primary repair. Ultrasound Obstet Gynecol. 2010;36:270.
- Shek K, Guzman Rojas R, Dietz HP. Residual defects of the external anal sphincter are common after OASIS repair. Neurourol Urodyn. 2012;31:913-14.
- Byrd LM, Hobbiss J, Tasker M. Is it possible to predict or prevent third degree tears? Colorectal Disease. 2005;7:311-318.
- Faridi A, Willis S, Schelzig P, Siggelkow W, Schumpelick V, Rath W. Anal sphincter injury during vaginal delivery – an argument for cesarean section on request? J. Perinat. Med. 2002;30:379-387.
- Glazener C, Abdalla M, Stroud P, Naji S, Templeton A, T. Russell I. Postnatal maternal morbidity: extent, causes, prevention and treatment. BJOG. 1995;102:282-287.
- Sultan Ah, Kamm MA, Hudson CN, Thomas JM, Bartram CI. Anal sphincter disruption during vaginal delivery. NEJM. 1993;329:1905-11.
- Oberwalder M, Connor J, Wexner SD. Meta- analysis to determine the incidence of obstetric anal sphincter damage. Br J Surg. 2003;90:1333-1337.
- Faltin D, Boulvain M, Irion O, Bretones S, Stan C, Weil A. Diagnosis of Anal Sphincter Tears by Postpartum Endosonography to Predict Fecal Incontinence. Obstetrics & Gynecology. 2000;95:643-647.
- Chaliha C, Sultan A, Bland J, Monga A, Stanton SL. Anal function: effect of pregnancy and delivery. Am J Obstet Gynecol. 2001;185:427-432.
- Zetterström J, Mellgren A, Jensen LL, et al. Effect of Delivery on Anal Sphincter Morphology and Function. Diseases of the Colon & Rectum. 1999;42:1253-1260.
- Steensma A, Burger C, Schouten W. Is dynamic investigation of the anal sphincter complex useful for assessment of the anal sphincter complex? Ultrasound Obstet Gynecol. 2008;32:397.
- Peschers UM, Delancey JO, Schaer GN, Schuessler B. Exoanal ultrasound of the anal sphincter: normal anatomy and sphincter defects. BJOG. 1997;104:999-1003.
- Valsky DV, Malkiel A, Savchev S, Messing B, Hochner-Celnikier D, Yagel S. 3D transperineal ultrasound follow-up of intrapartum anal sphincter tears and functional correlation with clinical complaints. Ultrasound Obstet Gynecol. 2007;30:449.
- Oom DMJ, West CR, Schouten WR, Steensma AB. Detection of anal sphincter defects in female patients with fecal incontinence: a comparison of 3D transperineal ultrasound and 2D endoanal ultrasound. Dis Colon Rectum. 2012;55:646-52.
- Anonymous. Rogers v Whitaker. Canberra: Commonwealth of Australia, 1992 (vol accessed 18.1.2014).
- Thomas V, Shek K, Guzman Rojas R, Dietz H. The latency between pelvic floor trauma and presentation for prolapse surgery. Ultrasound Obstet Gynecol. 2013;42:39.
- Brown S, Lumley J. Maternal health after childbirth: results of an Australian population based survey. BJOG. 1998;105:156-161.
- Johnstone SJ, Boyce PM, Hickey AR, Morris-Yatees AD, Harris MG. Obstetric risk factors for postnatal depression in urban and rural community samples. Australian & New Zealand Journal of Psychiatry. 2001;35:69-74.
- Kovacs G, Heath P, Heather C. First Australian trial of the birth- training device Epi-No: A highly significantly increased chance of an intact perineum. ANZJOG. 2004;44:347-348.
- Shek K, Langer S, Chantarasorn V, Dietz H. Does the Epi-No device prevent levator trauma? A randomised controlled trial. Int Urogynecol J. 2011;22:1521-8.






Leave a Reply