Heterotopic pregnancy describes a concurrent intrauterine and extrauterine ectopic pregnancy.1 It is a rare diagnosis, with an incidence estimated to be 1 in 30 000 spontaneous pregnancies.2 In pregnancies conceived using assisted reproductive therapies (ART), the incidence increases to 1 in 3900.3 Furthermore, caesarean section pregnancy (CSP) is also uncommon, occurring in less than 1 in 2000 pregnancies in women with previous caesarean delivery.4 5
There is limited evidence to guide management of heterotopic pregnancy. Medical treatment involves using localised or systemic methotrexate, while surgical encompasses procedures to remove the pregnancy tissue such as uterine wedge resection or hysterectomy. A recent study by Ramkrishna et al6 has shown that the use of intra-sac potassium chloride (KCl) and methotrexate is a successful intervention for management of non-tubal ectopic pregnancies, especially for women wishing to preserve fertility. Clearly, medical management is appropriate only in clinically stable cases.7 Surgical management is otherwise required.8
Case description
A 35-year-old woman, gravida 2 para 1, presented to hospital with vaginal bleeding and pelvic pain at 6 weeks gestation. This pregnancy was planned and spontaneously conceived. Her past obstetric history was of an emergency caesarean section at term, two years prior. She was clinically well and underwent a transabdominal and transvaginal ultrasound that diagnosed a probable early dichorionic-diamniotic (DCDA) pregnancy.
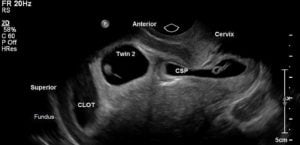
Figure 1. Sagittal view of the cervix and uterus demonstrating fundal clot, twin 2 intrauterine pregnancy and the caesarean scar pregnancy.
Repeat ultrasound at 7 weeks gestation demonstrated a live heterotopic DCDA pregnancy, with one gestational sac developing in the caesarean scar, and the other pregnancy was developing within the uterine cavity.
Initial management options that were considered included termination of the entire pregnancy with intra-sac and systemic methotrexate, termination of the CSP using ultrasound guided intra-sac KCl, with expectant management of the intrauterine pregnancy (IUP), or expectant management with inpatient observation and ultrasound guidance. The unpredictable nature of this pregnancy, with risks of severe haemorrhage, morbidity and mortality, was conveyed to the patient; however, she initially preferred to preserve the IUP and decided to proceed with intra-sac KCl to the CSP.
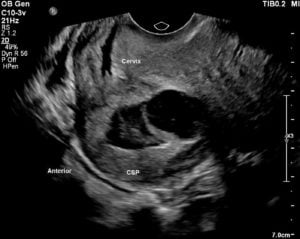
Figure 2. Sagittal view of the cervix and lower uterus demonstrating cervical canal and CSP distending anterior uterine serosa at the level of the caesarean scar. The volume of the CSP had increased subsequent to the KCl injection procedure.
Figure 2 shows the pre-intervention ultrasound. Using a 20-gauge needle, 2 mL (30 mmol/mL) intra-sac KCl was injected into the CSP using a transplacental entry. Asystole of the CSP was observed.
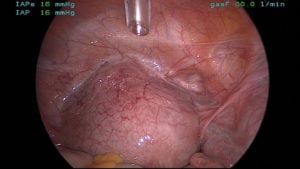
Figure 3. Uterovesical pouch concealing the CSP.
Repeat transvaginal ultrasound 10 days post selective reduction of the CSP revealed that the trophoblastic ring of the CSP had enlarged in size, with the trophoblast covered by serosa only, and no visible embryo. The large portion of the haematoma described previously was seen to be bypassing the CSP and reaching the internal os. Twin 2 remained live, now at a growth consistent with 9 weeks plus 1 day. Given the ongoing expansion of the CSP trophoblast and the potential risk of future uterine rupture with growth of the IUP, the patient was counselled to proceed with surgical treatment.
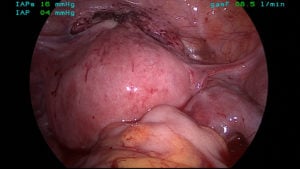
Figure 4. Surgical dissections exploring the CSP.
On the 17th day of the patient’s admission, the 14th day post elective KCl injection, operative laparoscopy was performed to excise the CSP. After precise and delicate surgical entry onto the uterovesical space and reflection of the bladder peritonium, diathermy was used to create surgical plane proximal to the bulging CSP and a wedge resection was made to remove the CSP using toothed graspers and suction. The defect was then closed in layers using delayed absorbable sutures. No excision of myometrium was required. A cervical dilatation and suction curettage were performed under laparoscopic vision to remove the IUP. Blood loss was moderate due to the vascularity of the gravid uterus; however, the patient remained stable postoperatively and did not require a red cell transfusion. She was discharged home three days later and over the following five weeks her βHCG was tracked down to zero.
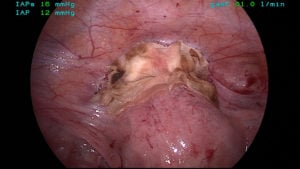
Figure 5. Following the surgical removal of the CSP with applied absorbable adhesive barrier.
Discussion
CSP in heterotopic pregnancy is a rare diagnosis. The number of previous caesarean section deliveries is not correlated to the likelihood of developing CSP, according to Kirk et al.9 There is a lack of evidence to recommend an ideal management approach and this makes clinical decision making difficult and non-uniform. These cases are clearly challenging to clinicians and distressing for patients. Therefore, multidisciplinary input, particularly from experts in ultrasound and advanced gynaecological surgery, is essential to effective and safe management of these complex cases.
Multidisciplinary input from senior clinicians, including subspecialty experts in laparoscopic surgery and gynaecological ultrasound, were involved in this patient’s care, with frequent and in-depth clinical meetings occurring in which this case was discussed and options for management explored. The ultimate decision to proceed with surgical management was challenging, as we respected our patient’s desires to conserve the IUP and had explored all conservative management options. Due to the persistent expansion of the ectopic sac, it was felt that on balance, surgical intervention was required to prevent uterine rupture and the resultant significant maternal morbidity.
A review of the literature demonstrates less than 30 published cases of CSP and concurrent IUP. Salomon et al10 described the first case of a heterotopic caesarean scar pregnancy successfully treated with KCl in the first trimester, with the patient having a 36-week emergency caesarean section for premature rupture of membranes. There are two reported cases in the literature describing successful IUP outcome after surgical management of the CSP. Demirel et al11 described a case of a heterotopic caesarean scar pregnancy managed with laparoscopic removal of the ectopic mass. They opted for surgical removal of the placental tissue and repair of the myometrium in light of the risks of weakening the lower uterine segment with use of medical management when coexisting with an IUP.12 The IUP continued without complication, with a healthy baby born at 38 weeks gestation via caesarean section.13 The other reported case in the literature also describes successful outcomes for the IUP after surgical excision of the CSP with utilisation of laparotomy as their surgical method of choice.14 In a case report, Gupta et al15 described suction aspiration as the therapeutic choice in their patient who wanted a successful pregnancy outcome and was also keen to preserve her fertility.
Accordingly, management of heterotopic pregnancy can be medical or surgical. Minimally invasive approaches, such as ultrasound-guided intra-sac injection of methotrexate, have been shown to be associated with high rates of pregnancy termination, without rupture, which may be especially useful in women wishing to preserve fertility.16 However, this management option is not always appropriate for use in cases of heterotopic pregnancy due to the adverse effects on the IUP. Cases of heterotopic pregnancies with implantation in the caesarean scar are rare, with most heterotopic ectopic pregnancies occurring in the tube. In the more common scenario of the heterotopic pregnancy involving a tubal ectopic, surgical management via salpingectomy is most favourable, as surgical termination of the extrauterine pregnancy can occur, while preserving the IUP.17 Yet, from the reports discussed here, consideration of other modalities are beneficial when the extrauterine pregnancies are in unusual locations not easily amenable to surgical intervention, and which may predispose to uterine rupture, such as at the caesarean scar.
Conservative management of CSP is also an option for consideration in certain isolated cases; however, it is often not a clinically preferred option due to the risks of uterine rupture. A recent study by Timor-Tritsch et al18 determined that both placental implantation in CSP and early (second trimester) placenta accreta share common histopathological features, likely representing different stages in the continuum leading to morbidly adherent placenta in the third trimester. These conditions all implant over the previous caesarean scar and can result in serious obstetric complications.19
Most heterotopic pregnancies are diagnosed at five to eight weeks gestation; however, there are reported cases of diagnosis as late as 35 weeks gestation.20 Delayed diagnosis and management of heterotopic pregnancies can be associated with significant maternal morbidity and mortality, and endanger the IUP. Commonly, the imaging modality of choice for diagnosis of heterotopic pregnancy is transvaginal ultrasonography. Recent literature has concluded that early transvaginal ultrasound performed by an experienced clinician is associated with a sensitivity of 92.4 per cent and a specificity of 100 per cent for detection of heterotopic pregnancy.21
This case contributes to the literature regarding the management of CSP as a heterotopic pregnancy. Ultrasound-guided intra-sac KCl may be considered for selective reduction of a CSP when preservation of a coexisting IUP is desired; however, this case demonstrates the need to offer surgical management for those who have failed non-surgical management. Early diagnosis of heterotopic pregnancy is essential to improve the possibility of preserving the IUP and reduce maternal morbidity and mortality, and transvaginal ultrasound adds to diagnostic accuracy. Clearly, with increasing prevalence of advanced maternal age, increased uptake of ART and rising caesarean section rates, clinicians may encounter more cases of heterotopic and CSP. Diagnosis and management of these complex cases should be individualised with appropriate counselling and careful assessment of the patient and a multidisciplinary approach.
References
- Govindarajan MJ, Rajan R. Heterotopic pregnancy in natural conception. Journal of Human Reproductive Sciences. 2008;1(1):37-8.
- Glassner MJ, Aron E, Eskin BA. Ovulation induced with clomiphene and the rise of heterotopic pregnancies: A report of two cases. J Reprod Med. 1990;35:175-8.
- Farnaghi S, Kothari A. Heterotopic pregnancy: a report of two cases. Australasian Journal of Ultrasound in Medicine. 2015;16(1):30-6.
- Jurkovic D, Hillaby K, Woelfer B, et al. First-trimester diagnosis and management of pregnancies implanted into the lower uterine segment Cesarean section scar. Ultrasound Obstet Gynecol. 2003;21,220-7.
- Seow KM, Cheng WC, Chuang J, et al. Methotrexate for Caesarian scar pregnancy after in vitro fertilization and embryo transfer. A case report. J Reprod Med. 2000;45,754-7.
- Ramkrishna J, Kan GR, Reidy KL, et al. Comparison of management regimens following ultrasound diagnosis of nontubal ectopic pregnancies: a retrospective cohort study. BJOG. 2017. https://doi.org/10.1111/1471-0528.14752.
- Fatema, N, Al Badi MM, Rahman M, Elawdy MM. Heterotopic pregnancy with natural conception; a rare event that is still being misdiagnosed: a case report. Clinical Case Reports. 2016;4(3):272-5.
- Fatema, N, Al Badi MM, Rahman M, Elawdy MM. Heterotopic pregnancy with natural conception; a rare event that is still being misdiagnosed: a case report. Clinical Case Reports. 2016;4(3):272-5.
- Kirk E, Condous G, Bourne T. Non-surgical management of ectopic pregnancy. Ultrasound Obstet Gynecol. 2006;27(1):91-100.
- Salomon LJ, Fernandez H, Chauveaud A, et al. Successful management of a heterotopic caesarean scar pregnancy: potassium chloride injection with preservation of the intrauterine gestation: case report. Hum Reprod. 2003;18:189-91.
- Demirel LC, Bodur H, Selam B, et al. Laparoscopic management of heterotopic cesarean scar pregnancy with preservation of intrauterine gestation and delivery at term: case report. Fertil Steril. 2009;91(4):1293.
- Demirel LC, Bodur H, Selam B, et al. Laparoscopic management of heterotopic cesarean scar pregnancy with preservation of intrauterine gestation and delivery at term: case report. Fertil Steril. 2009;91(4):1293.
- Demirel LC, Bodur H, Selam B, et al. Laparoscopic management of heterotopic cesarean scar pregnancy with preservation of intrauterine gestation and delivery at term: case report. Fertil Steril. 2009;91(4):1293.
- Armbrust R, Kratschell R, Henrich W, David M. Operative therapy for heterotopic scar pregnancy and successful birth of the intrauterine foetus – case report and review of the literature. Geburtshilfe Frauenheilkd. 2015;75(4):384–8.
- Gupta R, Vaikousi E, Whitlow B. Heterotopic caesarean section scar pregnancy. Journal of Obstetrics and Gynaecology, 2010;30(6):626-7.
- Ramkrishna J, Kan GR, Reidy KL, et al. Comparison of management regimens following ultrasound diagnosis of nontubal ectopic pregnancies: a retrospective cohort study. BJOG. 2017. https://doi.org/10.1111/1471-0528.14752.
- Wang C, Chen C, Wang H, et al. Successful management of heterotopic caesarean section scar pregnancy combined with intrauterine pregnancy after in vitro fertilisation-embryo transfer. Fertil Steril. 2007;88:13-16.
- Timor-Tritsch IE, Khatib N, Monteagudo A, et al. Cesarean Scar Pregnancies. Journal of Ultrasound in Medicine, 2015;34 601-10.
- Timor-Tritsch IE, Khatib N, Monteagudo A, et al. Cesarean Scar Pregnancies. Journal of Ultrasound in Medicine, 2015;34 601-10.
- Talbot K, Simpson R, Price N, Jackson SR. Heterotopic pregnancy. J Obstet Gynaecol. 2011;31:7-12.
- Li XH, Ouyang Y, Lu GX. Value of transvaginal sonography in diagnosing heterotopic pregnancy after in-vitro fertilization with embryo transfer. Ultrasound Obset Gynecol. 2013;41:563-9.






Leave a Reply